
The compound F4P-NEt2 has this bonding type, in which the P atom uses sp3d hybrid orbitals. For example, Sib s- uses sp3d hybrid orbitals to form five Si-F bonds, and SiF62 uses sp3d2 hydrid orbitals to form six Si-F bonds.
In many silicon compounds, the d orbitals of Si contribute to the hybrid orbitals and Si forms more than four 2c-2e covalent bonds. Unlike carbon, the valence shell of the silicon atom has available d orbitals.
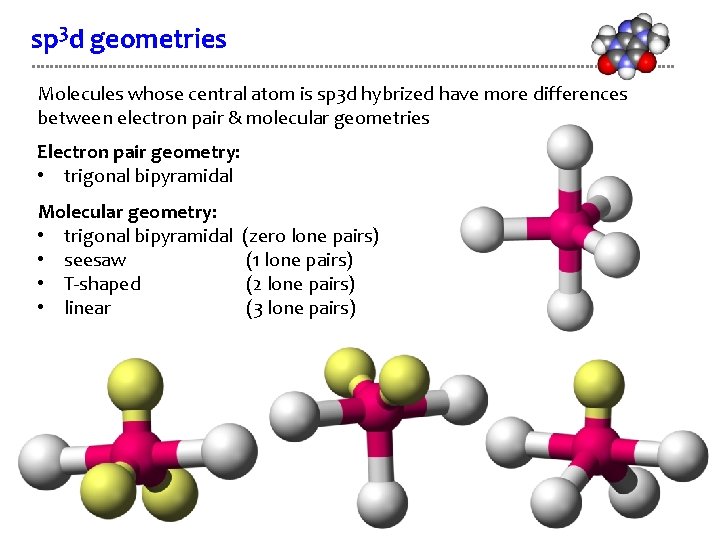
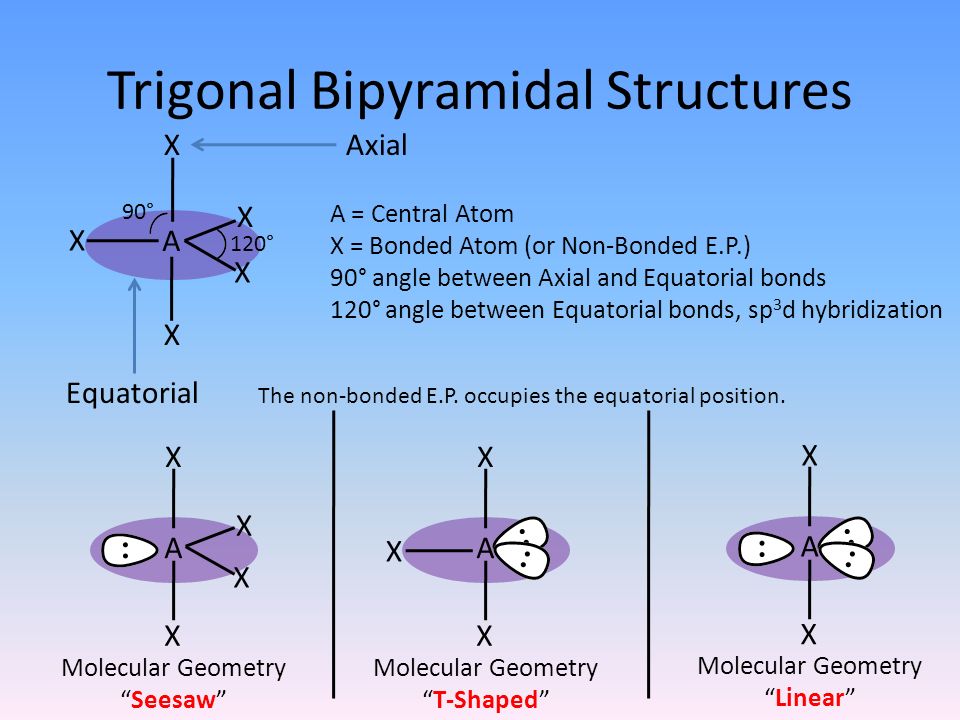
Three of the hybrid orbitals lie in a plane at angles of 120°, with the remaining two orbitals perpendicular to the plane, one above and one below. Hybridization of five atomic orbitals occurs by a combination of one s orbital, three p orbitals, and one d orbital, giving five sp3d hybrid orbitals in a trigonal bipyramidal arrangement (Figure 7.12). įIGURE 7.12 The five sp3d hybrid orbitals and their trigonal bipyramidal geometry. įIGURE 3.20 One of the five sp3d hybrid orbitals, and their five directions, that may be formed when d-orbitals are available and we need to reproduce a trigonal bipyramidal arrangement of electron pairs. Thus, by using sp3d hybridization, we end up with the same see-saw molecular geometry for the cation as that predicted by VSEPR theory (when the lone pair on Br is ignored). The four Br-F bonds in the cation are then formed by the overlap of the half-filled sp3d hybrid orbitals of the bromine atom with the half-filled p-orbitals of the terminal fluorine atoms. In this scheme, one of the sp3d hybrid orbitals is filled while the remaining four are half-occupied. It turns out that only sp3d hybridization will provide the necessary trigonal bipyramidal distribution of electron pairs around the bromine atom. Next we must select a hybridization scheme for the Br atom that is compatible with the predicted shape. Diagrams showing the directions of the maxima of octahedral sp3d2 and trigonal bipyramidal and square pyramidal sp3d hybrid orbitals. Thus, these are the only feasible structures that are symmetrical.Sp3d hybrid orbitals Figure 2. Again, the pentagonal bipyramidal structure is more stable. When we proceed in a similar manner for sp $^3$d $^3$, we have two options: heptagonal planar (bond angle $51.43°$) and pentagonal bipyramidal (bond angles $72°$ and $90°$. Of the two, trigonal bipyramidal is obviously a more stable structure (bond angles $90°$ and $120°$) than pentagonal planar (bond angle $72°$) ( $3$D here). The instant we do that, we are left with only two options: trigonal bipyramidal and pentagonal planar. Therefore, for symmetry about the plane, we must keep a third bond on the plane. We cannot put $2$ bonds on one side and $1$ bond on the other side, as that would be unsymmetrical ( $3$D here). Using the condition above, we have to arrange $3$ bonds around the plane, such that they are symmetric about it. Obviously, the two bonds form a plane ( $3$D here): I'm going to show that there are no better structures satisfying this condition for sp $^3$d and sp $^3$d $^3$ hybridisation.įor sp $^3$d, we're going to first arrange any two bonds at some angle (unknown). Note that this condition is satisfied by trigonal planar, tetrahedral, trigonal bipyramidal, octahedral and pentagonal bipyramidal geometry. Why are these arrangements preferred in favor of one with a simple symmetric arrangement of orbitals (which, I think, would minimize the repulsion)?įor a structure to be perfectly symmetric, it must satisfy this condition:įor any two bonds, the plane passing through them must divide the molecule into two symmetric halves. A molecule in which the central atom is $sp^3d^2$ hybridized ($\ce$) with five orbitals crammed into the equatorial plane and two sticking out above and below it.
